That group also supplied nearly all the patients surveyed for the study.
Spinal cord stimulators are small computers wired into the nerves,often to treat chronic pain. Advocates claim they are effective and low-risk;critics argue they are expensive and point to studies suggesting they and are.
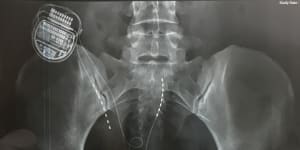
An X-ray showing a spinal cord stimulator in situ.Supplied
The paper,released by peak body Painaustralia last Wednesday,surveyed 73 Australian stimulator users. “The consumers sampled in this study were overwhelmingly positive about their experience of their spinal cord stimulator,” it concluded.
A page from the Spinal Cord Stimulator Implants Consumer Experience Report,December 2023.Supplied
It includes three positive case studies,including a wheelchair-bound man who was able to walk again. “When I woke up,I opened my eyes and thought my dreams have come true,” he says in the report.
Some 86 per cent of the patients surveyed were supplied through the Neuromodulation Society of Australia and New Zealand. The society also funded the report.
The society lists on its website. All four manufacture spinal cord stimulators.
Dr Chris Hayes,foundation director of Hunter Integrated Pain Service,said he resigned from Painaustralia’s board,partly as a result of the report’s publication.
Speaking as a clinician,he toldThe Age:“I do think there are some positives:it discloses the source of funding,the selection bias,and it refers to the two Cochrane studies that conclude this may be an ineffective treatment that comes with significant risks.
“But I am concerned the report could be misunderstood by vulnerable people who may not read the fine detail and gain the overall impression this is a very effective treatment.”
Painaustralia declined to answer questions about how much the Neuromodulation Society of Australia and New Zealand had paid for the report,nor if Painaustralia had received other funding from spinal cord stimulator manufacturers.
But Painaustralia’s website contains a fact sheet about spinal cord stimulation that advises the procedure is effective,minimally invasive,and should be considered early in a patient’s treatment. It was produced with funding from a company that makes spinal cord stimulators.
“As a not-for-profit organisation,we rely on funding from a range of sources. We are not influenced by external organisations. They do not have editorial input and we ensure transparency,” said Painaustralia CEO Giulia Jones.
“Participants[in the report’s survey] were a combination of those contacted by their pain specialist or choosing to respond to our social media call-out. As a cohort of 73 participants,this does not represent the experience of everyone who gets a spinal cord stimulator.”
The report was published on December 20,two days after Australian researchers arguing the stimulator industry was trying to use its financial resources to bury critical science.
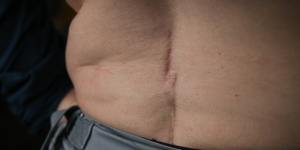
Lou Whelan shows the scarring from having two spinal cord stimulators implanted.Eddie Jim
“The report looks like yet another attempt by corporate interests to smother independent research,” said lead author Dr Adrian Traeger from the University of Sydney. “It’s a disservice to people with chronic pain. Very disappointing.”
Professor Chris Maher,founding director of the Institute for Musculoskeletal Health,said the “design and conduct of[the report] are both so flawed that even if there was no industry funding,you would just ignore this report”.
He pointed instead to a randomised controlled trial,(Journal of the American Medical Association),that found stimulators were no more effective than a placebo for chronic back pain after surgery. “People should believe the clinical trial,” he said.
The Neuromodulation Society of Australia and New Zealand describes the risks that come with a spinal cord stimulator as. The society did not reply to requests for comment by deadline.
But in the United States,spinal cord stimulators have the of medical device injury reports made to the Food and Drug Administration.
Australian data is patchy,and cannot be used to calculate absolute risks. But last year revealed 520 serious complications had been reported to the Therapeutic Goods Administration,leaving some people incontinent or unable to walk.
Two recent Cochrane reviews – generally considered gold-standard evidence – concluded the stimulators were for treating chronic back pain,or.
That has led some researchers,along with private health insurers,to call for a.
Fascinating answers to perplexing questions delivered to your inbox every week..